Claims substantiation
We are committed to working closely with you through the clinical trial and claims support process from study design and trial initiation through data analysis and interpretation. We are experienced in all aspects of project management and supervision with the ability to monitor and audit all aspects of the evaluation process.
Through our innovative global affiliations and by employing a vast array of the latest cutting edge technologies, we are able to take special care in protocol development for both standard studies as well as those that require creatively addressing your specific claims. Through consultation, execution and results interpretation, our goal is to assist in allowing your products to be uniquely positioned in an ever changing market.
Customized protocols are developed based on a client’s special needs and can include support for claims for the following specific target regions.
Hand and Body
Anti Stretch Marks, Anti Dimples, Firmness/Elasticity, Exfoliating, Hair Removing Efficacy, Slimming/Contour, Uplifting and Volume of Bust, Anti Aging Hands, Nail Health, Hot Room Clinical Testing for Deodorant and Antiperspirant (FDA and/or Clearcast methods)
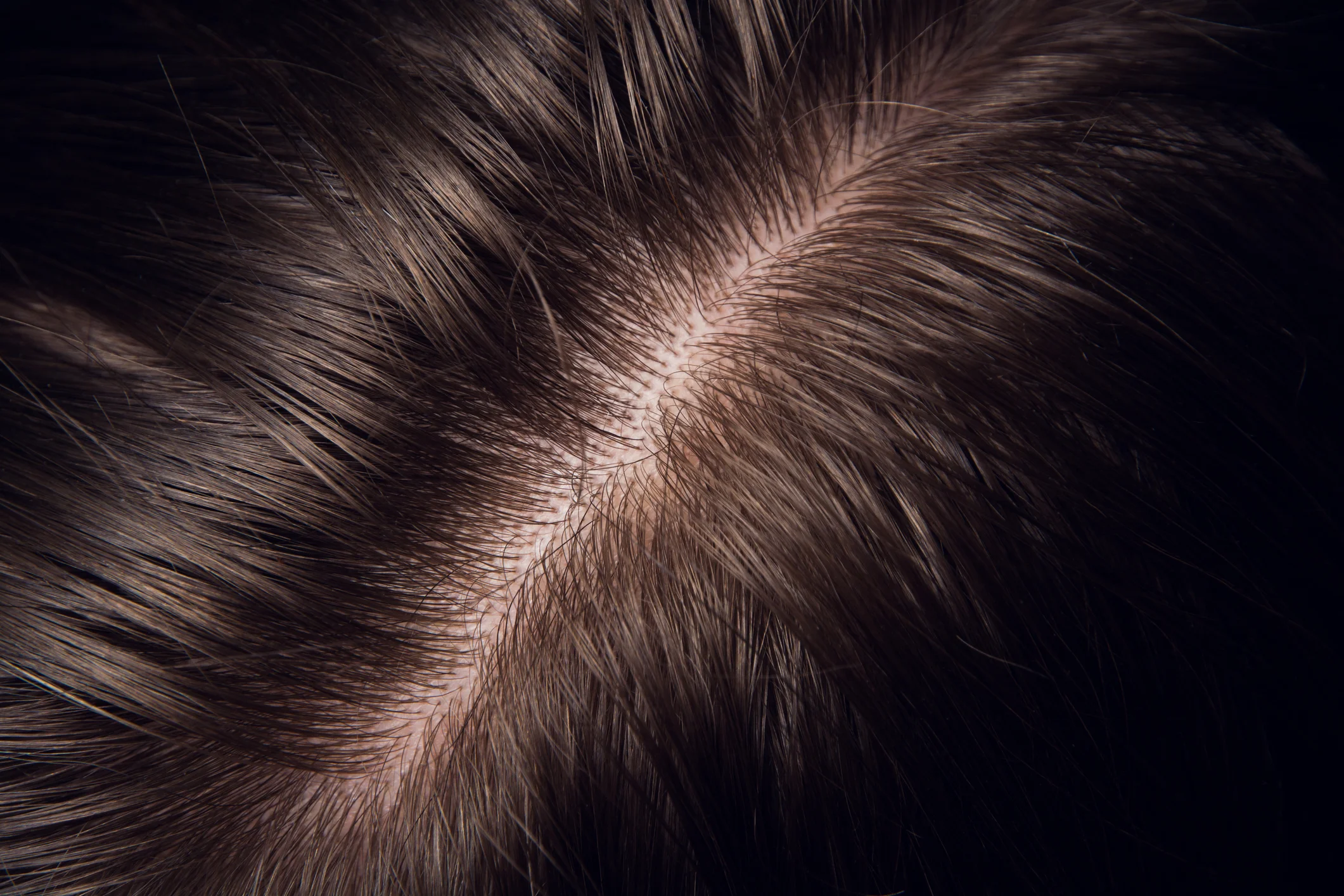
Hair and Scalp Care
Anti Sebum, Anti Hair Loss, Combing Effect, Duration of Color, Hair Growth, Smoothing Effect, Sheen, Softness, Strengthening Effect, Hydration, Scalp Health
Face
Anti Aging, Anti Dark Spots, Anti Sagging Effect, Anti Sebum Effect, Anti Under Eye Bags, Anti Wrinkle, Astringent Effect/Exfoliation, Cell Renewal, Complexion Homogeneity, Complexion Radiance, Elasticity/Firmness, Moisturizing/Hydration, Self Tanning, Lifting, Skin Brightening, Redness, Epidermal Melanin, Collagen Content
Sunscreen (SPF) and UVA
FDA 2011 21 CFR Parts 201 and 310 – Labeling and Effective Testing; Sunscreen Drug Products for Over-the-Counter Human Use – Food and Drug Administration (U.S.): Static, Water Resistant 40 Minute/Water Resistant 80 Minute (Water Resistant Testing includes Static determination)
ISO 24444 – Cosmetics – Sun protection test methods – In Vivo determination of the Sun Protection Factor (SPF). Current method provides for Static determination only.
International Sun Protection Factor; International Global Harmonization Method (SPF) [CTFA-SA, COLIPA, JCIA, PCPC]: Static, Water Resistant 40 Minute/Very Water Resistant 80 Minute (Water and Very Water Resistant Testing include Static determination)
AS/NZS 2604:2012 – Australian/New Zealand Standard: Static, Water Resistant 40 Minute, Water Resistant 80 Minute, 2-hour, 3-hour, 4-hour (Water Resistant Testing includes Static determination)
UVA In-Vivo Protection Efficacy:ISO 24442 – Cosmetics-Sun Protection Test Methods – In-Vivo Determination of Sunscreen UVA Protection, JCIA Method – Japan Cosmetics Industry Association (PA, PFA, PPD)
In-Vitro Test Methods
FDA 2011 21 CFR Parts 201 and 310 – Labeling and Effective Testing; Sunscreen Drug Products for Over-the-Counter Human Use – Food and Drug Administration (U.S.) Critical Wavelength – Necessary for Broad Spectrum claims
International Standard ISO 24443 – Determination of sunscreen UVA Photoprotection In-Vitro
COLIPA – “COLIPA In-Vitro UV Protection Method” – In-Vitro method for the determination of the UVA Protection Factor and Critical Wavelength values of sunscreen products, 2011
The Boots Star Rating – “Measurement of UVA:UVB Ratios According to the Boots Star Rating System – 2011 revision”
AS/NZS 2604:2012 – “Australian/New Zealand Standard; Sunscreen Products – Evaluation and Classification”